Volac Calf Volostrum - 450g
A colostrum substitute, high in protein and energy recommended for use when adequate supplies of good quality colostrum are unavailable. When mixed with 0.5 litresof warm water it will make up 1 calf feed and should be given as soon as possible after birth, preferably within 6 hours.
£ 16.50Cepravin DC Herd Pack 1X200
Recommended for routine dry cow therapy to treat existing sub-clinical infections and to prevent new infections which occur during the dry period.
£ 335.00Lamb volostrum 500g
500g of volostrum powder with 25g scoop. Provides 10 feeds as a natural alternative or 20 feeds as a natural supplement
£ 28.00Ectofly Pour On For Sheep
A clear blue coloured non-aqueous solution. For the treatment and control of headflies and the treatment of ticks and biting lice in sheep. For the prevention and treatment of blowfly strike in sheep. The active ingredient is cypermethrin high CIS 80:20. Target Species:Sheep
£ 50.00Farm Cam Safe & Efficent Farming
FarmCam Safe & Efficent Farming Easy to use wireless camera to keep an eye on your animals from the comfort of your own home, as well as being viewed on your mobile device.
£ 420.00Farm Cam Ip
FarmCam IP is a smart camera system that keeps you connected to critical events and locations on your farm wherever you are. A wireless camera system that connects to your WiFi router and works out of the box – view and zoom HD video directly on your smartphone, tablet, or computer through the FarmCam app. Installation takes just 5 minutes.
£ 220.00Infrared heatlamp & 175w bulb
Heatlamp Infrared 5m cable & dimmer plus Philips Infrared 175W Clear ES Screw Fit Heat Lamp Bulb - PAR38 bundle offer Heatlamp infrared assembly C/W 5m cable & dimmer.Screw fit, 175 watt infra red bulb. PAR38 (121 mm diameter) *while stock lasts
£ 22.00
Pen Strep 100ml
Pen & Strep Injection is indicated for use in cattle, horses, pigs and sheep for the treatment of infections caused by, or associated with, organisms sensitive to Penicillin and/or Streptomycin Product Name: Pen/Strep Price (Ex VAT): £17.00 Pack: 100ml Vet Prescription Required: Yes Legal Category: POM-V Available for sale to ROI: No
£ 17.00Allopurinol Tabs 300mg 1x28
for conditions associated with too much uric acid in the body, such as kidney disease, metabolic disorders, certain skin diseases, cancer ect. Product Name: Allopurinol Tabs 300mgPrice (Ex VAT): £4.00Pack: 1x28Vet Prescription Required: YesLegal Category: POM-VAvailable for sale to ROI: No
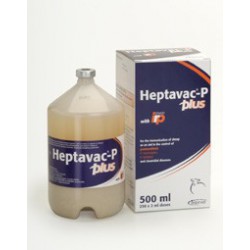
£ 4.00Heptavac-P Plus
Pasteurella pneumonia is the biggest killer of sheep. Nearly all sheep carry the organisms on their tonsils and when animals are stressed, disease often results.Product Name: Heptavac-P PlusPrice (Ex VAT): £38.00Pack: 50mlVet Prescription Required: NoLegal Category: POM-VPSAvailable for sale to ROI: No
£ 38.00Engemycin Spray 200ml
Product Name: Engemycin Spray Price (Ex VAT):£9.00Pack: 200ml Vet Prescription Required: Yes Legal Category: POM-V Available for sale to ROI: No
£ 9.00Norodine 24 Injection 100ml
Norodine 24 is indicated in the treatment of acute, subacute and chronic conditions of bacterial origin in horses, cattle, pigs, dogs and cats. Product Name: Norodine 24 Injection Price (Ex VAT): £20.50 Pack: 100ml Vet Prescription Required: Yes Legal Category: POM-V Available for sale to ROI: No
£ 20.50Alamycin Aerosol 140 gm
Alamycin aerosol is a cutaneous spray indicated for the treatment of foot rot in sheep and topical infections caused by oxytetracycline-sensitive organisms in cattle, sheep and pigs. Product Name: Alamycin Aerosol Price (Ex VAT): £9.00 Pack: 140 gm Vet Prescription Required: Yes Legal Category: POM-V Available for sale to ROI: No
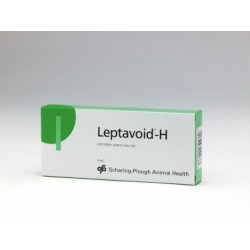
£ 9.00Leptavoid-H
For the active immunization of cattle from 1 month of age against Leptospira interrogans serovar hardjo and Leptospira borgpetersenii serovar hardjo.Product Name: Leptavoid-HPrice (Ex VAT): £26.40Pack: 20ml/10 doseVet Prescription Required: NoLegal Category: POM-VPSAvailable for sale to ROI: No
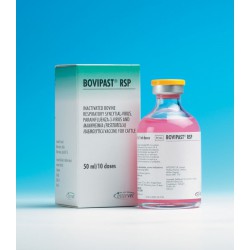
£ 68.50Bovipast RSP
An aqueous suspension for subcutaneous injection.Product Name: Bovipast RSPPrice (Ex VAT): £72.00Pack: 50ml/10 doseVet Prescription Required: YesLegal Category: POM-VAvailable for sale to ROI: No
£ 72.00Program 80 Cat Injection
For the six-month long-term prevention and control of flea infestation and the treatment of flea allergic dermatitis in cats. Product Name: Program 80 Cat InjectionPrice (Ex VAT): £65.00Pack: 1x1Vet Prescription Required: YesLegal Category: POM-VAvailable for sale to ROI: No
£ 65.00Chloromed 150mg/g
Calves: This product is indicated for the treatment of respiratory disease in calves caused by Pasteurella spp, sensitive to chlortetracycline. This product should be mixed thoroughly through their daily feed and should not be administrated prior to the feeding. Do NOT use in calves over 6 months
£ 24.00